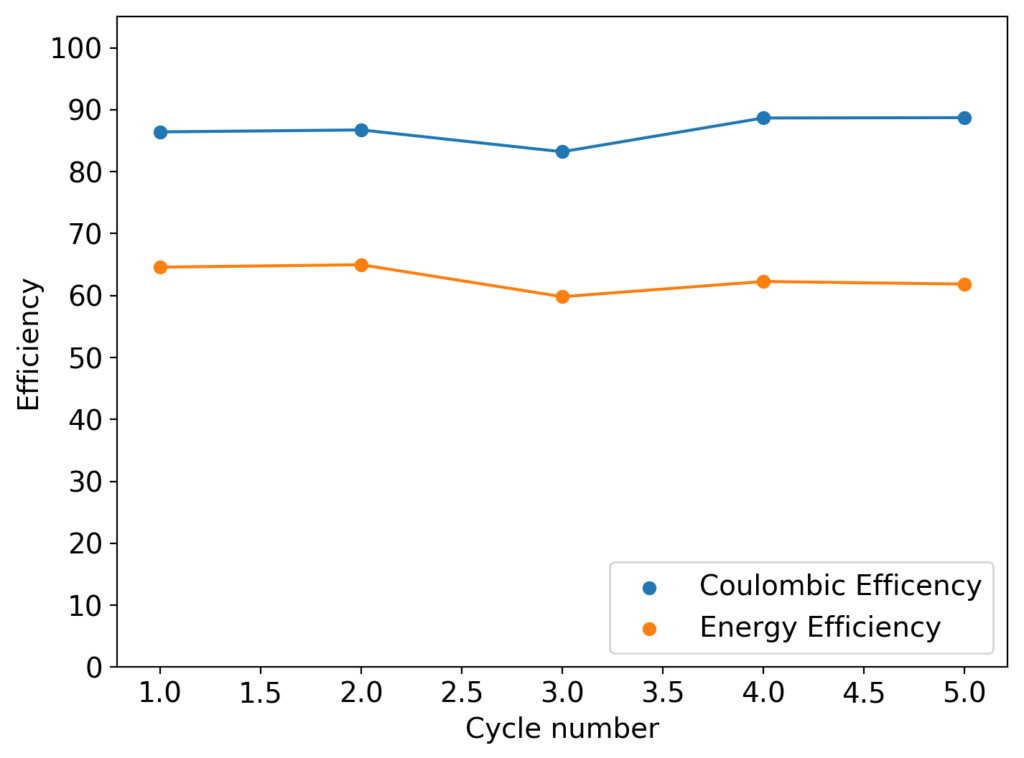
This past week I did not post any new results for Zn-Br batteries. This is because I started to face significant reproducibility issues in my spacer based batteries with no separator. The image below shows you some of the typical curves I was getting from my batteries using PEG-200 containing solutions at a ZnBr2 concentration of 3M with different NaCl or NaBr additions. The battery started just fine – with CE values close to 90% – but fell sharply thereafter, with big increases in series resistance follower by large losses.
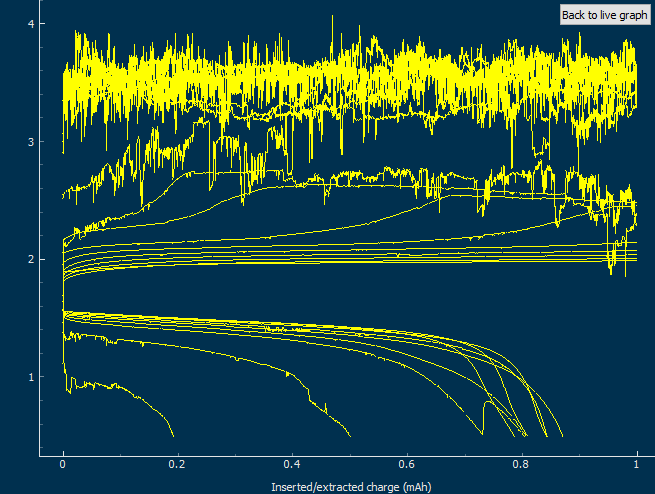
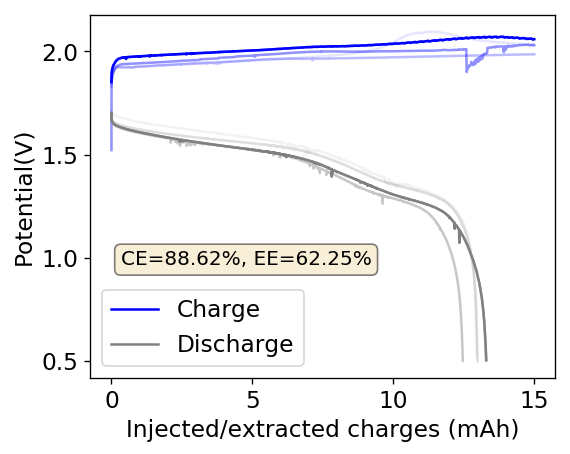
After a lot of investigation, the problem seems to be the adhesion of the Zn deposits to the anode’s graphite electrode. Even though the anode is always polished before every battery, the Zn deposits sometimes just “fall off” and – since there is no separator – that Zn falls to the cathode and is thereby lost and simply reacts slowly with the bromine. This was confirmed by moving again to a Zn metallic anode (0.2mm thickness) which didn’t show the above problems, as you can see in the curves below.
Although relatively normal CE and EE values were achieved for this battery configuration, dendrite formation was evident, both in the charge/discharge curves and after taking the battery apart (where dendrites were quite large). It is clear, both from NaCl and NaBr experiments, that additions of these supporting electrolytes contributes heavily to dendrite formation. It also seems pretty clear that going from 1% PEG-200 to 6% PEG-200 or higher doesn’t help enough with dendrite formation – they still form, even if a bit slower – but the heavy increase in series resistance is not worth the trade-off. If you try to add more PEG-200 and reduce the series resistance with NaBr or NaCl, then you just get the dendrites again.
From these experiments, it is now pretty clear why commercial ZnBr2 batteries do not use PEG-200 as an additive – at least in very large quantities – it might work to suppress formation of Zinc dendrites at lower ZnBr2 concentration (<1M) but at the concentrations required for energy density values greater than 30-40 Wh/L it just doesn’t seem to work well enough. Furthermore, while PEG-200 can be used with little effect in highly conductive KOH solutions that are used in some Zn chemistries (like Zn/Mn oxide batteries) it just doesn’t work when the electrolyte’s conductivity is significantly lower, such as is the case with ZnBr2 solutions.
All hope is not lost though. While PEG-200 by itself might not be able to prevent dendrites in this configuration, it is possible that low concentrations of PEG-200 plus other additives might have a synergistic enough effect to help us alleviate the problem. One such potential case is with the use of PEG-200 and Tween-20, which at 0.5% each, have shown to be both quite effective and synergistic at reducing Zinc dendrites. The experimentation continues!
Now I have to track down some Tween-20 and try that in the batteries I have been playing with 🙂
You are right about the resistance of the battery going up when adding PEG 200 (in my case PEG 400). I’m trialing a few different types of electrode in an attempt to help reduce the resistance of the battery.
For the cathode have tried using a graphite crucible, sodium silicate (water glass)with graphite, graphite rods pushed through carbon felt, just carbon felt and various types of copper mesh/stainless steel mesh with carbon filled HDPE coatings. The anodes are usually just copper/stainless steel mesh/foil with or without the carbon filled HDPE coatings. I have also tried just having the carbon felt on the top and bottom of the battery.
All of the batteries needed voltages too high to be useful when charging. Unless you are going to be pushing really low amps (milliamps), the charging voltage will be too high, and the voltage on discharge too low. Some of this is due to the reasons you mentioned above but also because the electrodes are too far apart (to avoid dendrites). I going to continue trying as many ways I can find to build ZnBr2 batteries while hopefully you find a way for the chemistry to work.
Cheers for all of the effort. Looking forward to your next post.
Thanks for your comment! Certainly, low energy efficiency is definitely a big problem with setups that have large electrode separation. The huge “batteries in a jar” with centimeters between electrodes that many people imagine making in a DIY manner will not be able to conduct easily enough for the energy efficiency to be acceptable. In my experiments the maximum electrode separation to achieve higher energy efficiencies (>60%) is around 3mm, anything above that and the voltaic loses become way too high for any significant current density to be used. For low separation you then absolutely need to avoid dendrites, as a dendrite of just 3mm is enough to destroy the battery.
Your point about Zinc oxide formation in the anode is also very valid and I believe something I have observed myself when these batteries require voltages above 2V (when water splitting becomes a very important process). Something worth noting is also that elemental bromine intercalates normal graphite and dramatically lowers its conductivity, increasing the potential of the cathode if a graphite conductor is used. Graphitic felts can solve this problem, but then you need a titanium electrode to collect current as graphite gets continuously damaged by the intercalation of the halide.
Thanks for the tip about the intercalation of the graphite and bromine. It might explain what happened with my crucible battery which started off brilliantly but by the next day began to need higher charging voltages to to receive the same amount of current. The anode of the battery was fine. The crucible was also too porous so the stainless steel tie straps I used to hold the copper wire onto the outside of the crucible started to corrode.
Once the titanium foil I ordered arrives I’ll be testing it in the various battery forms I have been using. In spite of the dendrites I’m still going to build a battery with a narrow electrode separation to see what current I can pull. I expect I’ll only get one to two charges before it fails but I want to see what I can get regardless.
Cheers.